Average Atomic Mass Worksheet. The abundances of all isotopes should add as much as 100% .The isotope Ag-107 has an abundance of fifty one.86%. The term “average atomic mass” is a __weighted_______________________average, and so is calculated in a special way, from a “normal” common. Isotopes are named after the “mass quantity,” or the sum of protons and neutrons in one atom. The abundance of all of the isotopes ought to add as much as 100%.
They also calculate the atomic mass of elements given the odds of every isotope. A primary presentation to introduce a variety of the terminology concerning atomic weight, atomic structure, and atomic number awaits your college students. An instance of the calculations finding common atomic mass for carbon is given, and a method of…

The different major isotope of Bromine has an atomic mass of 80 and a relative abundance of 49%. If you should discover the typical atomic mass of an element, you will want to search for the atomic mass and the abundance of every isotope in that element.
College Students Additionally Viewed
The narrator shows viewers the means to calculate the typical atomic mass utilizing the relative abundances… In order to read or download common atomic mass worksheet show all work answers e book, you should create a FREE account.

As lengthy as you employ atomic mass items in your average mass calculation, however, the two values are numerically equivalent. In this element worksheet, learners build a periodic table as a category. Each student is assigned three elements and should create a paper diagram indicating the factor name, the symbol, the atomic quantity, the common atomic mass and different…
How To Find Common Atomic Mass
The trainer will supply M&M’s, Skittles and Reeses Pieces to provide information about mass and abundance. Some isotopes are unstable, or radioactive, which means they may decay over time and switch into one other isotope or secure factor. In the interactive simulation, participants manipulate parts to create isotopes and collect data….
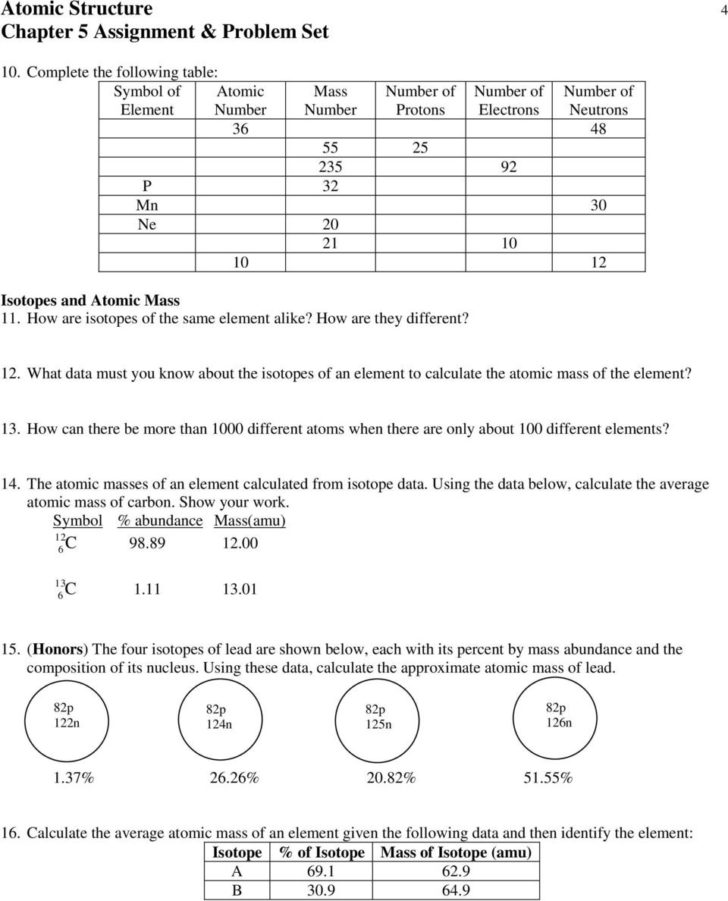
The mass quantity for each isotope is the sum of numbers of protons and neutrons within the nucleus. Each proton and every neutron weigh 1 atomic mass unit . In this isotopes worksheet, students calculate an average atomic mass of specific elements.
Average Atomic Mass
Just select your click then download button, and complete a suggestion to begin downloading the e book. If there is a survey it only takes 5 minutes, try any survey which works for you.

Each isotope contributes proportionally to its abundance . You can find this in the same supply you discovered the mass. The abundances of all isotopes ought to add up to 100 percent .The isotope Ag-107 has an abundance of 51.86%.
Have you ever questioned why the atomic mass listed on the periodic table is not a complete number? This video explains how weighted averages are calculated and relates the common to the relative atomic mass.

In this lots of atoms worksheet, students complete a chart of seven isotopes with their isotope symbol, atomic mass, atomic quantity, and the variety of protons, neutrons and electrons. They calculate the typical atomic mass of two components. Solve the thriller of these awkward atomic masses that include decimals with a video from JFR Science.
Atomic masses are nearly all the time written in phrases of atomic mass items , generally called the dalton . Never add a different unit of mass after the quantity without changing it.

Through the activity, blossoming chemists perform calculations on various isotopes, as represented by beans and legumes, to acquire the average… Did you know that physique armor actually incorporates uranium? Learn about how isotopes of the identical element have totally different masses and distinctive bodily properties.

1 atomic mass unit is defined as 1/12 the mass of one carbon-12 atom. The only distinction between two isotopes of the identical factor is the variety of neutrons per atom, which impacts the atom’s mass. By specializing in “Candium”, pupils can work via this information to study extra about different isotopes.

Ag-109 is slightly less widespread with an abundance of forty eight.14%. This means that a typical sample of silver is 51.86% Ag-107 and 48.14% Ag-109.
In this common atomic mass worksheet, college students read about how the protons and neutrons make up the atomic mass of a component. They find the typical atomic mass given four problems. For this isotopes worksheet, college students discover the common atomic mass given the lots of two isotopes.

This is a fast method to examine whether your answers make sense. One isotope of Bromine has an atomic mass of 78.92u and a relative abundance of 50.69%. The abundance tells you the way widespread the isotope is, as a proportion of all atoms of the element.
A lab investigation models a fictional factor utilizing pasta. The setup uses three totally different pasta sorts to characterize three different isotopes.

Pupils decide the common mass of various beans as they carry out a collection of aggressive experiments. Calculate the typical atomic mass for every element based mostly on the pure abundance of its isotopes. With uncommon exceptions, components later on the periodic table have the next common mass than the weather before it.

Include your email tackle to get a message when this question is answered.

As an atomic construction reference and evaluation tool, this handout fits the invoice. The first page provides definitions and tables of orbitals, electrons, and power ranges. The second page is a chance to practice determining numbers of…
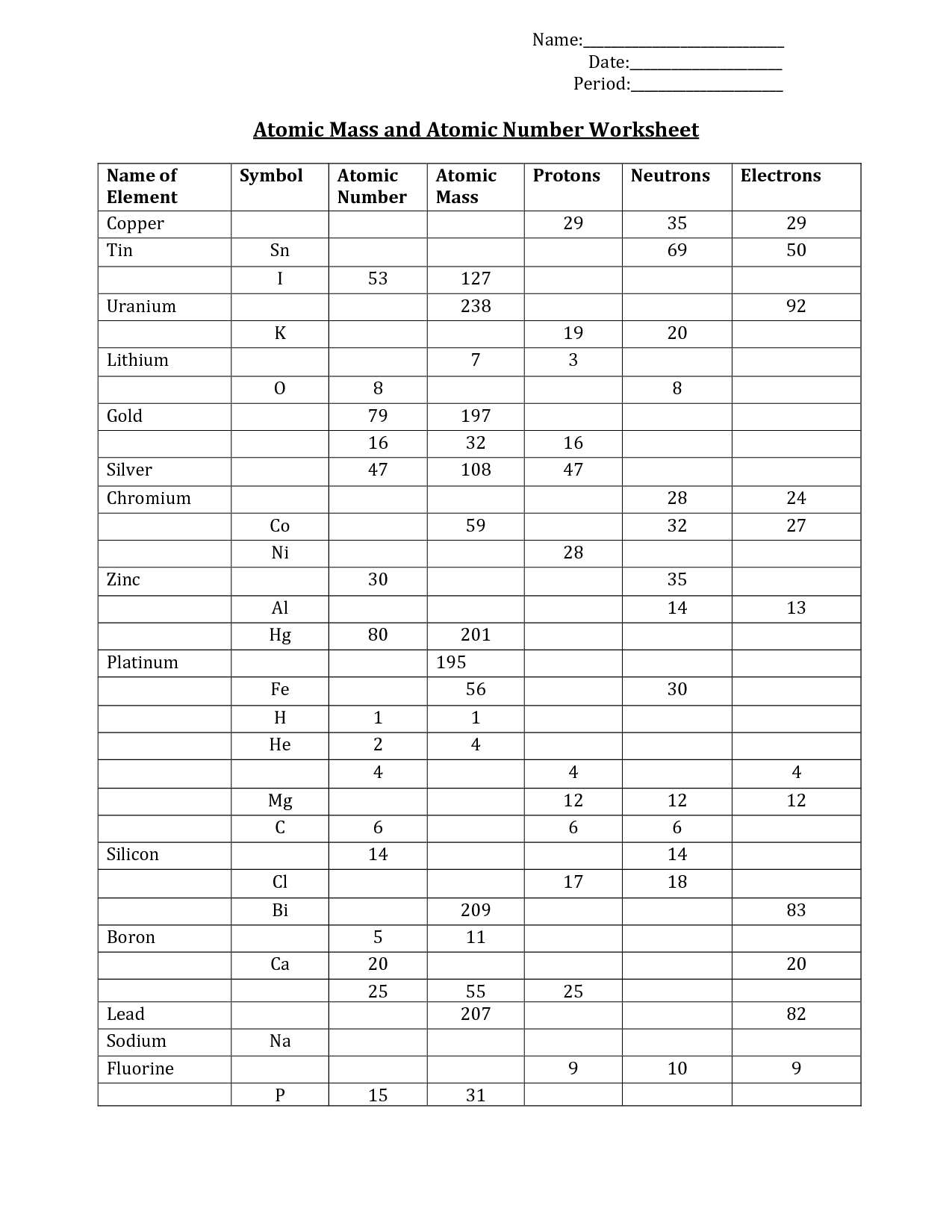
In this atomic construction and periodic desk worksheet, students full a desk with isotopes, their atomic masses, the variety of protons, neutrons and electrons and mass number. They also discover the average atomic mass of atoms and… Using a chart of student test grades for instance, curious chemists learn how to calculate weighted averages.

This means Ag-109 has two more neutrons per atom than Ag-107, giving it slightly extra mass. Sometimes modeling is the best approach to working with microscopic particles.

Instead, it is the common mass per atom for a typical sample of a given component. If you could measure the mass of billions of particular person atoms, you would calculate this worth the same means you’d discover any average.

Displaying all worksheets related to – Average Atomic Mass Calculations. Displaying all worksheets associated to – Average Atomic Mass.

They apply this information to elements on the periodic desk. As apply in calculating average atomic lots, learners…

The average atomic mass is often written beneath the element image. High schoolers discover the common atomic mass of M & M’s. In this chemistry lesson, students are given a pattern of the component chocolatium and the atomic mass of each pattern.

The time period relative atomic mass is sometimes used as a synonym for average atomic mass. There is a slight distinction for the reason that relative atomic mass has no items; it is a measure of mass relative to the carbon-12 atom.

“It taught me the easiest approach to calculate the common atomic mass of an element, and it is so comprehensible.” Relative molecular lots sure have a lot of decimal places! Watch the video to learn how the variety of particles in a single mole of carbon-12 determines relative atomic and molecular plenty.

Her research are targeted on proteins and neurodegenerative ailments. Isotopes are named after the “mass quantity,” or the sum of protons and neutrons in a single atom. Meredith Juncker is a PhD candidate in Biochemistry and Molecular Biology at Louisiana State University Health Sciences Center.
Educator Edition Save time lesson planning by exploring our library of educator reviews to over 550,000 open academic sources . Displaying all worksheets related to – Atomic Mass And Number. Ignore any isotopes that don’t have an abundance listed.



